which-interferences-impact-icp ms accuracy-most

ICP-MS is a sophisticated analytical technology that detects trace elements in diverse materials. However, its accuracy can be harmed by a variety of interferences that impair measurement precision. Understanding interferences is essential for increasing ICP-MS performance. By identifying the causes of these interferences, analysts can develop effective techniques to mitigate their effects. This blog discusses typical interferences in ICP-MS and their sources, as well as recommended procedures to reduce them, ensuring accurate and repeatable findings.
What Are the Common Interferences in ICP‑MS?
Spectral Interferences and Their Sources
Spectral interferences arise when ions from elements in the sample interact with those of interest during analysis. These interferences are often caused by polyatomic or molecular ions with the same mass-to-charge ratio (m/z) as the analyte. Common sources include argon-based species, such as argon dimers (Ar2+), which can imitate the signal of other elements, such as potassium. These interferences provide erroneous measurements, making it difficult to discern between components. To remedy this, ion source changes or the usage of reaction cells can be used to mitigate the effects of spectrum overlap.
Non‑Spectral Interferences Explained
Non-spectral interferences in ICP-MS impact ionization and detection without causing spectral overlap. These interferences are caused by matrix effects, in which components in the sample affect the target analyte’s ionization efficiency. For example, large amounts of one element might inhibit or amplify the ionization of another, skewing the results. Other non-spectral interferences include chemical processes that create undesired ions or isotopes. These interferences make it difficult to get precise results, necessitating procedures such as internal standardization or sample dilution to mitigate their effects.
Isobaric Overlaps and Mass Shifts
Isobaric overlaps and mass shifts occur when ions from distinct elements or isotopes have the same m/z value, leading to misleading results. ICP-MS detection can be affected by elements with similar masses, such as isotopes of various elements, causing interference. Mass shifts can also be generated by changes in ionization circumstances, which modify the m/z ratios of certain ions. These concerns are most common when elements have similar isotopic masses, such as calcium (40Ca) and titanium (48Ti). To successfully separate ions and correct for these interferences, high-resolution mass spectrometers or reaction/collision cells are required.
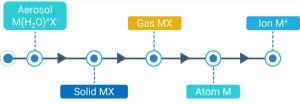
Effective Strategies to Minimize ICP‑MS Interferences
Collision/Reaction Cell Techniques
Collision and reaction cell approaches reduce interference in ICP-MS by using gas-filled cells that interact with ions. In a collision cell, ions collide with a gas, which reduces polyatomic interferences by fragmentation or energy loss. A reaction cell, on the other hand, uses reactive gases to selectively react with interfering species, converting them into non-interfering ions. These strategies enhance ICP-MS precision by eliminating spectrum overlaps and non-spectral effects, resulting in a cleaner signal and precise quantification of target elements, particularly in complex matrices.
Sample Preparation and Dilution Strategies
Proper sample preparation and dilution are critical to minimizing ICP-MS interferences. Diluting materials to lower concentrations reduces matrix effects and helps alleviate ionization suppression. Sample digestion procedures, such as acid digestion, can also break down complicated matrices, ensuring that analytes are identifiable. Filtration or centrifugation is commonly used to remove particle debris that might interfere. Furthermore, the inclusion of internal standards during sample preparation can help account for any changes in ionization and increase the reproducibility of findings, assuring accuracy even in difficult samples.
Choosing the Right Calibration Methods
Calibration is necessary for proper ICP-MS findings. Choosing the appropriate calibration procedure helps account for interferences that may alter the measurements. External calibration includes establishing a calibration curve using established concentration standards, whereas internal calibration employs an additional standard to account for any matrix or ionization effects. Multi-point calibration guarantees that the system stays linear across a wide range of concentrations, whilst matrix-matched calibration helps to mitigate the influence of sample matrix effects. Calibration enhances the precision and repeatability of ICP-MS readings, particularly for complex or unknown samples.
Conclusion
ICP-MS is an effective technique; however, its accuracy can be affected by interferences such as spectrum overlaps, non-spectral matrix effects, and isobaric overlaps. Understanding these interferences and their causes is critical for producing accurate findings. Analysts can mitigate the effects of these interferences by using procedures such as collision/reaction cells, good sample preparation, and effective calibration methods. Adopting these principles improves the precision and accuracy of ICP-MS analysis, leading to better decision-making in domains requiring trace element detection, including environmental monitoring, pharmaceutical research, and materials science.



